 Host Cell DNA Residual Detection Kit for HEK293(qPCR)
This kit is designed for the quantitative detection of the HEK293 Cell DNA in intermediate, semi-finished and finished products of various biological products.This kit adopts the principle of the Taq...
More+
Host Cell DNA Residual Detection Kit for HEK293(qPCR)
This kit is designed for the quantitative detection of the HEK293 Cell DNA in intermediate, semi-finished and finished products of various biological products.This kit adopts the principle of the Taq...
More+
 Mycoplasma DNA Detection Kit (qPCR)-ZY002
The kit is used to qualitatively detect the presence of mycoplasma contamination in master cell banks, working cell banks, virus seed lots, control cells, and cells for clinical therapy.The kit uses q...
More+
Mycoplasma DNA Detection Kit (qPCR)-ZY002
The kit is used to qualitatively detect the presence of mycoplasma contamination in master cell banks, working cell banks, virus seed lots, control cells, and cells for clinical therapy.The kit uses q...
More+
 Secret to new CAR T-cell therapy: ninja nanobodies
The NRC developed Canada’s first nanobody-based CAR T-cell therapy targeting CD22, offering a precise, innovative treatment for blood cancers with potential for broader applications.
More+
Secret to new CAR T-cell therapy: ninja nanobodies
The NRC developed Canada’s first nanobody-based CAR T-cell therapy targeting CD22, offering a precise, innovative treatment for blood cancers with potential for broader applications.
More+
 Contributing to the “Wuzhong Highland” in Biopharmaceutical Industry丨Hillgene Joined the “Innovative Pharmaceutical CXO Alliance in the Yangtze River Delta Region”
From October 13 to 16, 2021, the "First China (Suzhou) Taihu Pharmaceutical Innovation Conference" hosted by the Wuzhong District People's Government of Suzhou City and undertaken by the...
More+
Contributing to the “Wuzhong Highland” in Biopharmaceutical Industry丨Hillgene Joined the “Innovative Pharmaceutical CXO Alliance in the Yangtze River Delta Region”
From October 13 to 16, 2021, the "First China (Suzhou) Taihu Pharmaceutical Innovation Conference" hosted by the Wuzhong District People's Government of Suzhou City and undertaken by the...
More+
 Considerations on “IND Application Studies and Application Materials for Cellular Therapy Products”
In recent years, tumor immunotherapy technology has made rapid progress, and gene-modified cell products represented by chimeric antigen receptor modified T cells (CAR-T) have entered the clinic one a...
More+
Considerations on “IND Application Studies and Application Materials for Cellular Therapy Products”
In recent years, tumor immunotherapy technology has made rapid progress, and gene-modified cell products represented by chimeric antigen receptor modified T cells (CAR-T) have entered the clinic one a...
More+
 Solução CDMO para Vetores Lentivirais
Alta Produção: 2E11 TU/lote, escala 100L Alta Qualidade: 5E8 TU/mL, suspensão, livre de soro Alta Velocidade: 45 dias/lote
More+
Solução CDMO para Vetores Lentivirais
Alta Produção: 2E11 TU/lote, escala 100L Alta Qualidade: 5E8 TU/mL, suspensão, livre de soro Alta Velocidade: 45 dias/lote
More+
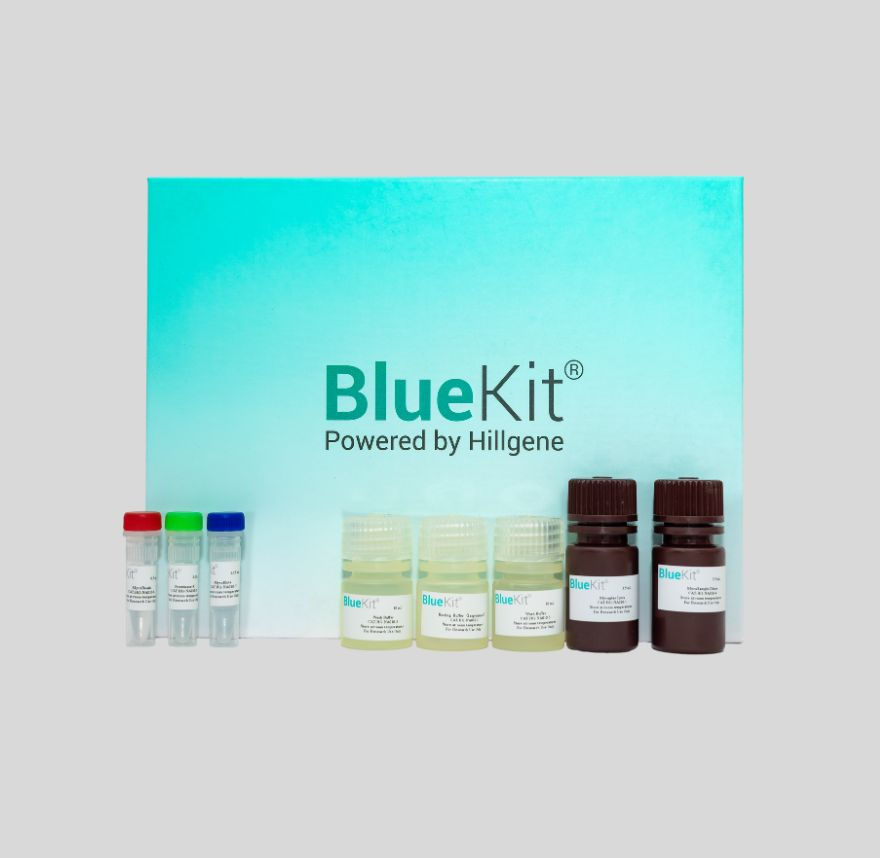 CD19 CAR-T Cell Preparation Kit
CD19 CAR T cell preparation kit is composed of three parts: T cell activation and expansion unit, T cell purification unit and CD19 CAR gene transduction unit. The T cell activation and expansion unit...
More+
CD19 CAR-T Cell Preparation Kit
CD19 CAR T cell preparation kit is composed of three parts: T cell activation and expansion unit, T cell purification unit and CD19 CAR gene transduction unit. The T cell activation and expansion unit...
More+
 Host Cell DNA Fragment Residual Detection Kit for HEK293(qPCR)
This kit is a kit specially designed for quantitative detection of the size distribution of residual HEK293 Cell DNA fragments in intermediates, bulk products and final products of various biological ...
More+
Host Cell DNA Fragment Residual Detection Kit for HEK293(qPCR)
This kit is a kit specially designed for quantitative detection of the size distribution of residual HEK293 Cell DNA fragments in intermediates, bulk products and final products of various biological ...
More+
 Good News | Hillgene Won the Title of “Star of Tomorrow” among the China Biopharmaceutical CDMO Companies in Star Awards-2021 BPID
On October 11-12, 2021, the BPID Biopharmaceutical Industry Development Conference and the 2021 Life and Health Industry Summit Forum were successfully concluded in Shanghai. The conference discussed ...
More+
Good News | Hillgene Won the Title of “Star of Tomorrow” among the China Biopharmaceutical CDMO Companies in Star Awards-2021 BPID
On October 11-12, 2021, the BPID Biopharmaceutical Industry Development Conference and the 2021 Life and Health Industry Summit Forum were successfully concluded in Shanghai. The conference discussed ...
More+
 Notification of “CAR-T Cell Quality Control Testing Research and Non-clinical Study Considerations”
Chimeric antigen receptor T cells (CAR-T) refer to the transfer of genetic material with specific antigen recognition domains and T cell activation signals into T cells through gene modification techn...
More+
Notification of “CAR-T Cell Quality Control Testing Research and Non-clinical Study Considerations”
Chimeric antigen receptor T cells (CAR-T) refer to the transfer of genetic material with specific antigen recognition domains and T cell activation signals into T cells through gene modification techn...
More+
 Regulation&Guideline(China)
Regulation&Guideline(China)NoRegulation&GuidelineIssued byRelease timeDownload01Technical Guideline for the Research of Human Cell Therapy and the Quality Control of Drug ProductsCDE2003/3/20doc02Con...
More+
Regulation&Guideline(China)
Regulation&Guideline(China)NoRegulation&GuidelineIssued byRelease timeDownload01Technical Guideline for the Research of Human Cell Therapy and the Quality Control of Drug ProductsCDE2003/3/20doc02Con...
More+