 Tips for Successful Lentiviral Transduction
Estimate IFU for MOI accuracy; use functional titration, optimize vectors, transfection, and enhancers to boost lentiviral efficiency.
More+
Tips for Successful Lentiviral Transduction
Estimate IFU for MOI accuracy; use functional titration, optimize vectors, transfection, and enhancers to boost lentiviral efficiency.
More+
 Live丨Writing a New Chapter for Cellular and Gene Therapies, Paving the Road to an Innovatively Developed and Industrialized Future
Cell and gene therapy technology is undoubtedly one of the most popular biotechnology fields at present, and is also regarded as the next generation of innovative therapies for disease treatment. In t...
More+
Live丨Writing a New Chapter for Cellular and Gene Therapies, Paving the Road to an Innovatively Developed and Industrialized Future
Cell and gene therapy technology is undoubtedly one of the most popular biotechnology fields at present, and is also regarded as the next generation of innovative therapies for disease treatment. In t...
More+
 The Comprehensive Affairs Department of NMPA Issued the Announcement on “Technical Guideline for the Pharmaceutical Study and Evaluation of Cellular Therapy Products” (No. 216, 2017)
In recent years, with the continuous development of basic theories, technical means and clinical medical exploration and research such as stem cell therapy, immune cell therapy and gene editing, cell ...
More+
The Comprehensive Affairs Department of NMPA Issued the Announcement on “Technical Guideline for the Pharmaceutical Study and Evaluation of Cellular Therapy Products” (No. 216, 2017)
In recent years, with the continuous development of basic theories, technical means and clinical medical exploration and research such as stem cell therapy, immune cell therapy and gene editing, cell ...
More+
 Solution CDMO de Vecteurs Lentiviraux
Production élevée : 2E11 TU/lot, échelle 100L Haute qualité : 5E8 TU/mL, suspension, sans sérum Haute vitesse : 45 jours/lot
More+
Solution CDMO de Vecteurs Lentiviraux
Production élevée : 2E11 TU/lot, échelle 100L Haute qualité : 5E8 TU/mL, suspension, sans sérum Haute vitesse : 45 jours/lot
More+
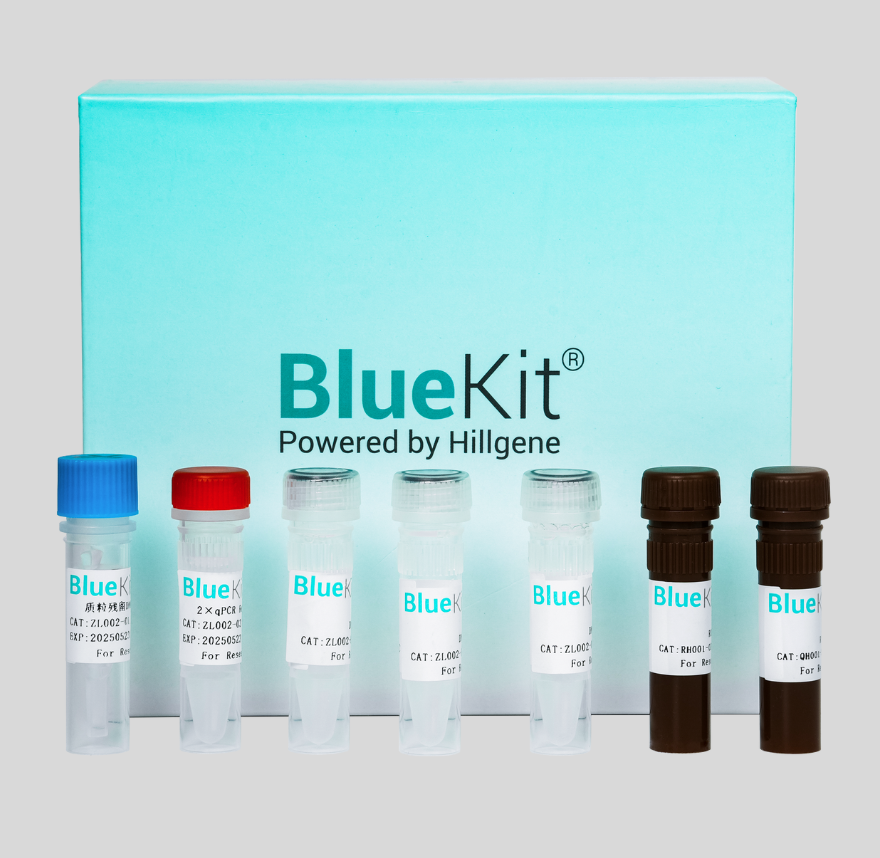
 Host Cell RNA Residual Detection Kit for Human(RT-qPCR)
This kit is designed for the quantitative detection of residual Human total RNA in various biological products to improve control quality of nucleic acid.This kit adopts the principle of the RT-PCR fl...
More+
Host Cell RNA Residual Detection Kit for Human(RT-qPCR)
This kit is designed for the quantitative detection of residual Human total RNA in various biological products to improve control quality of nucleic acid.This kit adopts the principle of the RT-PCR fl...
More+
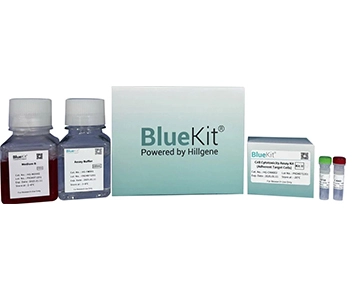
 Vaccinia Capping Enzyme ELISA Detection Kit
This kit uses double-antibody sandwich enzyme-linked immunosorbent assay (ELISA) method. Add Vaccinia Capping Enzyme standard and test samples to the microtiter plate precoated with anti-Vaccinia Capp...
More+
Vaccinia Capping Enzyme ELISA Detection Kit
This kit uses double-antibody sandwich enzyme-linked immunosorbent assay (ELISA) method. Add Vaccinia Capping Enzyme standard and test samples to the microtiter plate precoated with anti-Vaccinia Capp...
More+
 How to Choose a Cell Viability or Cytotoxicity Assay
Choose cell health assays based on your model, marker type, culture system, and timing needs; multiplex for richer, reliable data.
More+
How to Choose a Cell Viability or Cytotoxicity Assay
Choose cell health assays based on your model, marker type, culture system, and timing needs; multiplex for richer, reliable data.
More+
 Hillgene Reached a Strategic Cooperation with Biotheus in Climbing to New Heights in China for CAB-T Cell Therapy Products
On August 14, 2021, Jiangsu Spectrum Biopharmaceutical Co., Ltd. (hereinafter referred to as "Spectrum Bio") and Promis Biotech Co., Ltd. (hereinafter referred to as "Promis") reac...
More+
Hillgene Reached a Strategic Cooperation with Biotheus in Climbing to New Heights in China for CAB-T Cell Therapy Products
On August 14, 2021, Jiangsu Spectrum Biopharmaceutical Co., Ltd. (hereinafter referred to as "Spectrum Bio") and Promis Biotech Co., Ltd. (hereinafter referred to as "Promis") reac...
More+
 The Comprehensive Affairs Department of NMPA Issued the “Announcement on the Direct Reporting of Adverse Reactions by Marketing Authorization Holders (Draft for Comments)”
In order to implement the Opinions on Deepening the Reform of the Review and Approval System to Encourage Innovation in Drugs and Medical Devices (Office Letter [2017] No. 42) issued by the General Of...
More+
The Comprehensive Affairs Department of NMPA Issued the “Announcement on the Direct Reporting of Adverse Reactions by Marketing Authorization Holders (Draft for Comments)”
In order to implement the Opinions on Deepening the Reform of the Review and Approval System to Encourage Innovation in Drugs and Medical Devices (Office Letter [2017] No. 42) issued by the General Of...
More+
 Soluzioni CDMO per Vettori Lentivirali
Produzione Elevata: 2E11 TU/lotto, scala 100L Alta Qualità: 5E8 TU/mL, sospensione, senza siero Alta Velocità: 45 giorni/lotto
More+
Soluzioni CDMO per Vettori Lentivirali
Produzione Elevata: 2E11 TU/lotto, scala 100L Alta Qualità: 5E8 TU/mL, sospensione, senza siero Alta Velocità: 45 giorni/lotto
More+
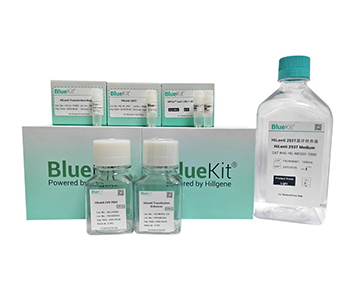
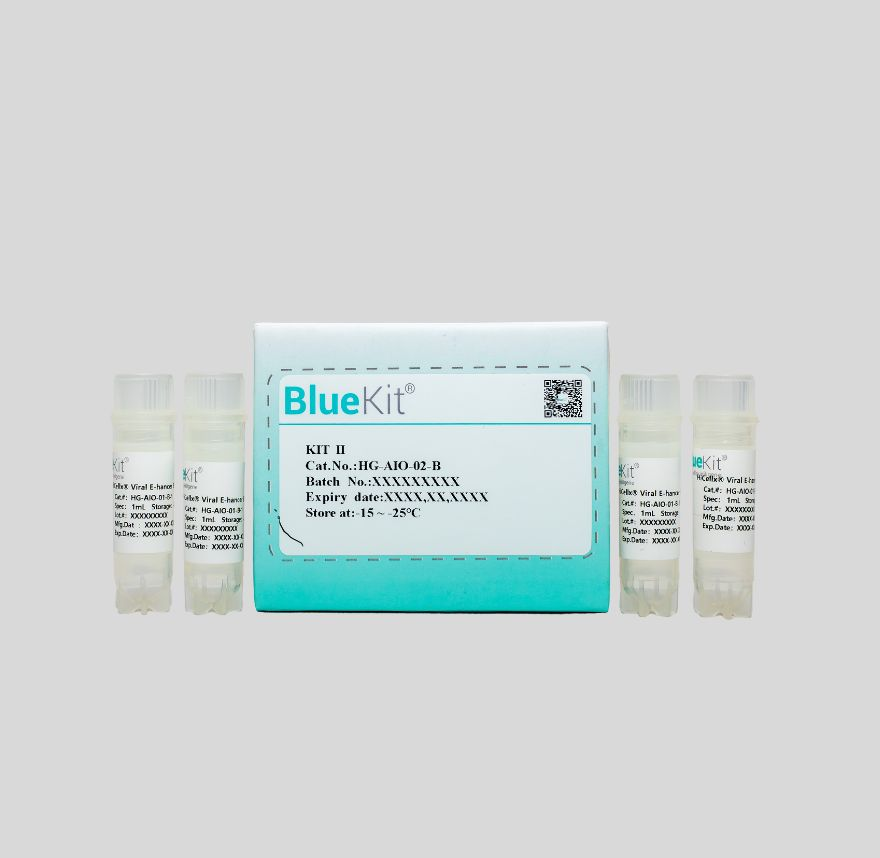
 Viral Transduction Enhancer A/B/C (ROU/GMP)
Hillgene's Viral E-hancer series products, as a class of highly efficient and non-cytotoxic lentiviral or retroviral transduction enhancers, are widely used in the viral transduction process of T ...
More+
Viral Transduction Enhancer A/B/C (ROU/GMP)
Hillgene's Viral E-hancer series products, as a class of highly efficient and non-cytotoxic lentiviral or retroviral transduction enhancers, are widely used in the viral transduction process of T ...
More+