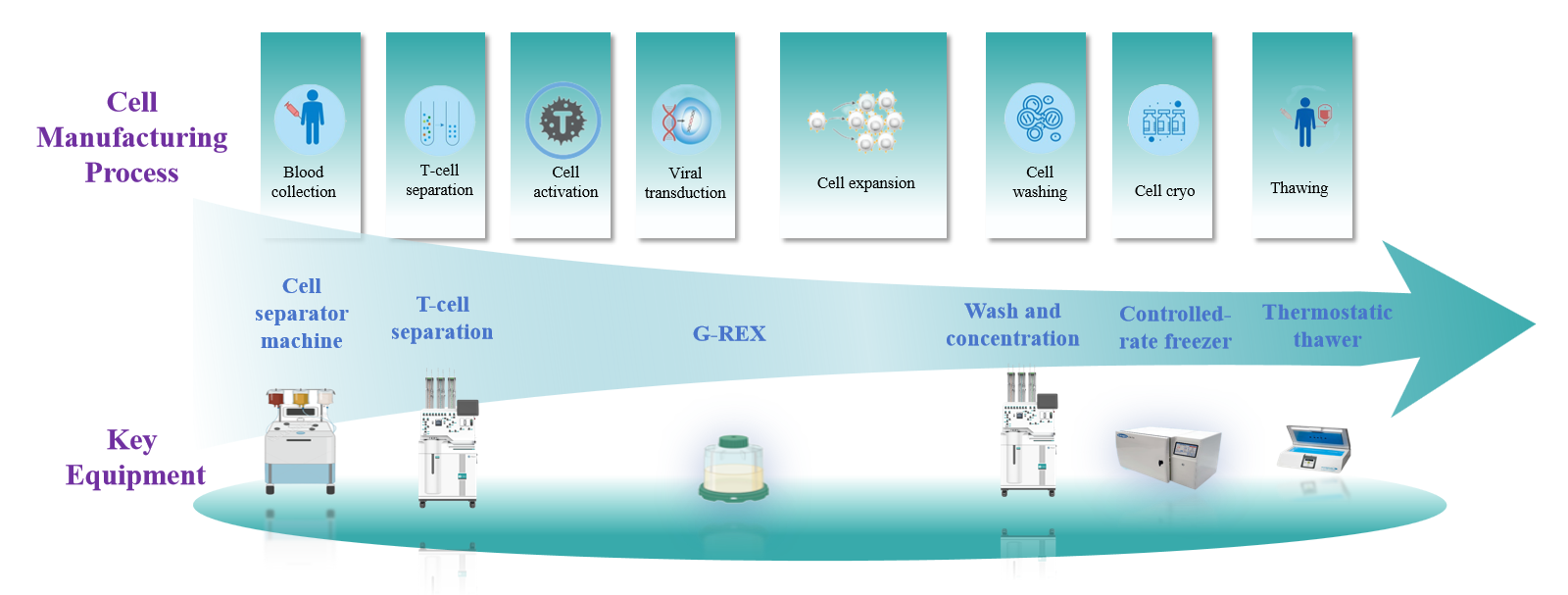
This kit is a comprehensive solution designed specifically for automated cell preparation platforms, integrating automated CAR-T production with end-to-end quality control. It provides standardized, closed-system, high-throughput support for BCMA CAR-T preparation and quality control in research settings. Fully compatible with closed automated systems, it significantly reduces variability caused by manual operations while enhancing experimental stability, batch-to-batch consistency, and data traceability. This product is intended for research use only and is not intended for clinical diagnosis, treatment, or in vitro diagnostics.
For Research Use Only.

Designed specifically for rapid release of CAR-T cell product
All unit components are validated to meet regulatory requirements
(1).jpg)