.webp) NK Kit
This kit is a comprehensive solution designed specifically for automated cell preparation platforms, integrating automated NK cell production with end-to-end quality control. It provides standardized,...
More+
NK Kit
This kit is a comprehensive solution designed specifically for automated cell preparation platforms, integrating automated NK cell production with end-to-end quality control. It provides standardized,...
More+
.webp) NK Manual Kit
The kit includes:Cell Cytotoxicity Assay KitCD3/CD56 Assay KitFastPure Cell Total RNA Isolation KitRT SuperMix for qPCR(+gDNA wiper) KitResidual K562 feeder cell Detection Kit
More+
NK Manual Kit
The kit includes:Cell Cytotoxicity Assay KitCD3/CD56 Assay KitFastPure Cell Total RNA Isolation KitRT SuperMix for qPCR(+gDNA wiper) KitResidual K562 feeder cell Detection Kit
More+
 Trypsin ELISA Detection Kit
This kit is designed for the quantitative detection of residual Trypsin content in intermediates,semi-finished products and finished products of various biological products by using a double antibody ...
More+
Trypsin ELISA Detection Kit
This kit is designed for the quantitative detection of residual Trypsin content in intermediates,semi-finished products and finished products of various biological products by using a double antibody ...
More+
![Hillgene Won the BioCon Awards [Annual CDMO Excellence Award] Hillgene Won the BioCon Awards [Annual CDMO Excellence Award]](/uploads/image/20250530/hillgene-won-the-biocon-awards-annual-cdmo-excellence-award.webp) Hillgene Won the BioCon Awards [Annual CDMO Excellence Award]
On September 6, BioCon Expo 2022, the 9th International Biopharmaceutical Conference and Exhibition, was held as scheduled at the Hangzhou International Expo Center. The BioCon Awards Ceremony and the...
More+
Hillgene Won the BioCon Awards [Annual CDMO Excellence Award]
On September 6, BioCon Expo 2022, the 9th International Biopharmaceutical Conference and Exhibition, was held as scheduled at the Hangzhou International Expo Center. The BioCon Awards Ceremony and the...
More+
 Newly Revised “Provisions for Drug Registration”
Chapter I General ProvisionsArticle 1 These Measures are formulated in accordance with the Drug Administration Law of the People's Republic of China (hereinafter referred to as the "Drug Admi...
More+
Newly Revised “Provisions for Drug Registration”
Chapter I General ProvisionsArticle 1 These Measures are formulated in accordance with the Drug Administration Law of the People's Republic of China (hereinafter referred to as the "Drug Admi...
More+
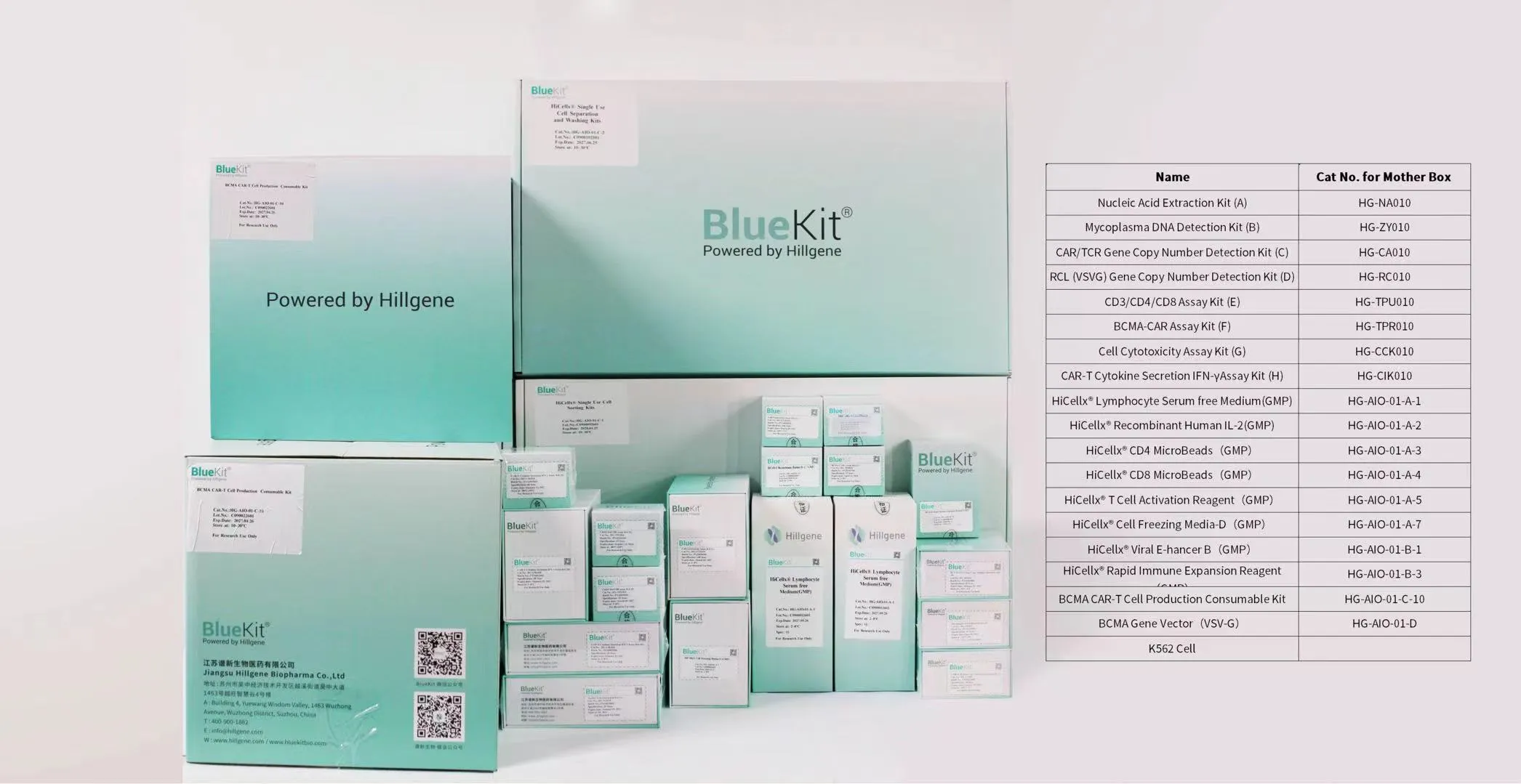 BCMA CAR-T Kit
Designed specifically for rapid release of CAR T-cell drugsAll unit components are validated to meet regulatory requirements
More+
BCMA CAR-T Kit
Designed specifically for rapid release of CAR T-cell drugsAll unit components are validated to meet regulatory requirements
More+
 Breaking | Hillgene Was Accepted as a Candidate of the “Unicorn” Enterprises Fostering Program
Science and technology are the primary productive forces. National and regional development cannot be separated from scientific and technological innovation. In order to implement the innovation-drive...
More+
Breaking | Hillgene Was Accepted as a Candidate of the “Unicorn” Enterprises Fostering Program
Science and technology are the primary productive forces. National and regional development cannot be separated from scientific and technological innovation. In order to implement the innovation-drive...
More+
 Notification of Public Comment Soliciting on “Technical Guideline for the Pharmaceutical Study and Evaluation of Cell-based Immunotherapy Products (Draft for Comments)”
In 2017, the former State Food and Drug Administration issued the "Technical Guidelines for Research and Evaluation of Cell Therapy Products (Trial)", which generally elaborated on the techn...
More+
Notification of Public Comment Soliciting on “Technical Guideline for the Pharmaceutical Study and Evaluation of Cell-based Immunotherapy Products (Draft for Comments)”
In 2017, the former State Food and Drug Administration issued the "Technical Guidelines for Research and Evaluation of Cell Therapy Products (Trial)", which generally elaborated on the techn...
More+