If CAR-T therapy is the “star” of the immunotherapy field, then natural killer (NK) cell therapy is poised to become the next “top trend.” With its three core advantages—readiness for use, safety, and broad-spectrum efficacy—NK cell therapy has attracted pharmaceutical companies worldwide to compete for a foothold in this space. The starting gun has fired, but no one has crossed the finish line yet. On the global NK cell therapy track, major pharmaceutical companies are sprinting full speed ahead, vying for the historic moment of “first-to-market.”
I. Global Competitive Landscape
In terms of clinical development, China leads the world with 62 trials (44%), followed closely by the United States with 45 (32%); Asian countries collectively account for over 61% of clinical trials. However, the vast majority of trials remain in early stages—Phase I trials account for 39.7%, and Phase I/II trials account for 30.5%, with no Phase III or IV trials yet initiated [1]. Overall, NK cell therapy remains in the early to mid-stage of clinical development, and the thorough validation of safety and efficacy remains the top priority.
Table 1. Overview of NK cell therapy drug pipelines that have entered Phase I/II clinical trials
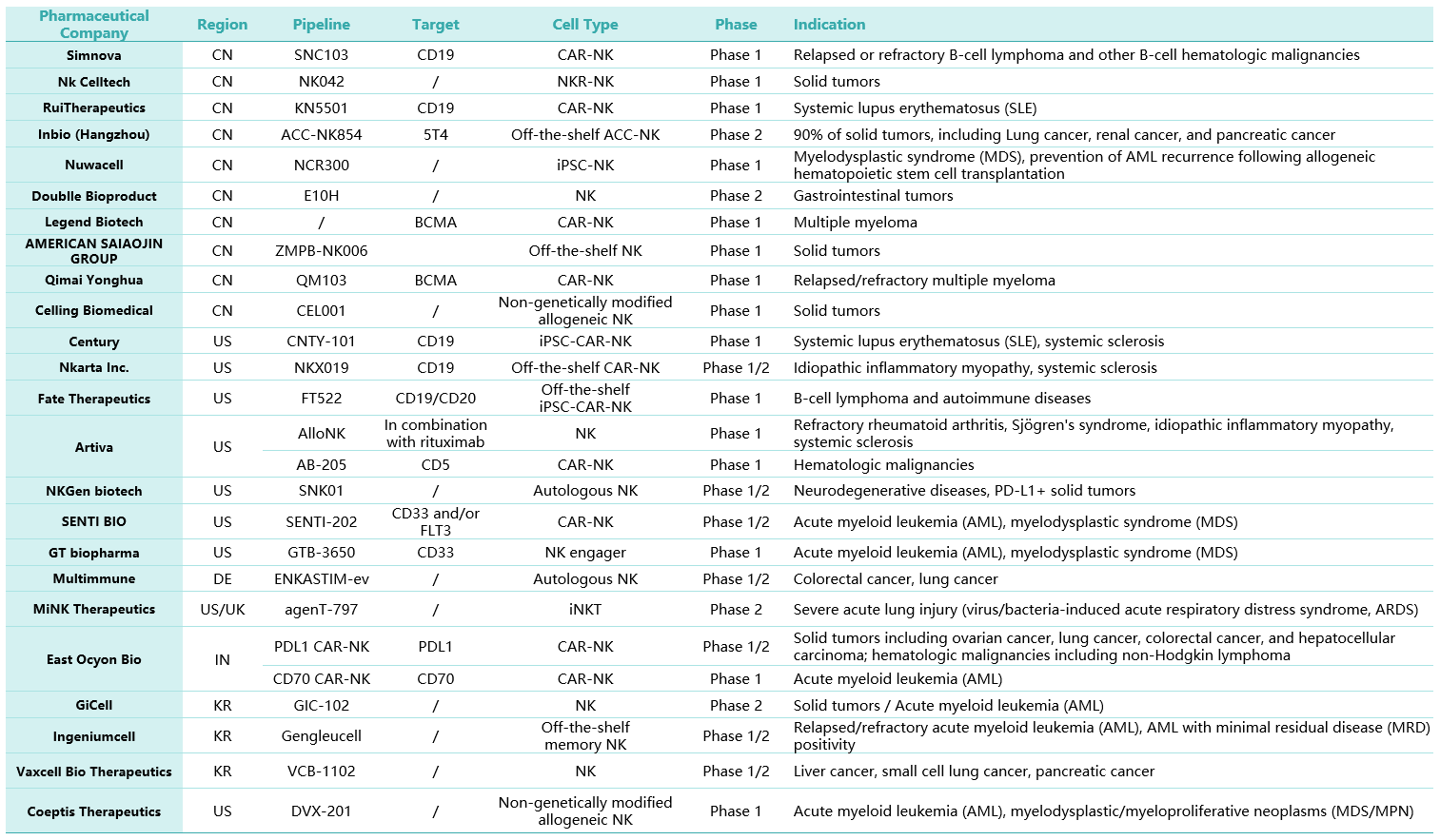
(Data sourced from internal research findings; partial display only)
II. Evolution of Targets and Indications
Hematologic malignancies: CD19 remains the most widely used core target for CAR-NK applications, primarily targeting B-cell lymphomas; BCMA, meanwhile, focuses on multiple myeloma and serves as the core target in this area.
Solid tumors: Target selection is becoming more diverse. Although the efficacy of NK cell therapy in solid tumors currently lags behind that in hematologic malignancies, positive signs are emerging: engineered CAR-NK cells can enhance tumor infiltration and resist microenvironmental suppression, and some patients have achieved objective responses following combination therapy. More importantly, they demonstrate excellent safety profiles, with severe cytokine release syndrome (CRS) being rare, laying the foundation for combination therapy and long-term administration. Their safety and engineering potential make them a promising option in the field of solid tumors.
Breakthroughs in Autoimmune Diseases: A 2025 study in The Lancet showed that allogeneic CD19 CAR-NK therapy for refractory systemic lupus erythematosus (SLE) achieved disease remission in 67% of patients, with only one case of mild cytokine release syndrome. CAR-NK is expanding from oncology into autoimmune diseases such as SLE and systemic sclerosis, emerging as a new frontier.
Figure 1. Common NK cell therapy strategies
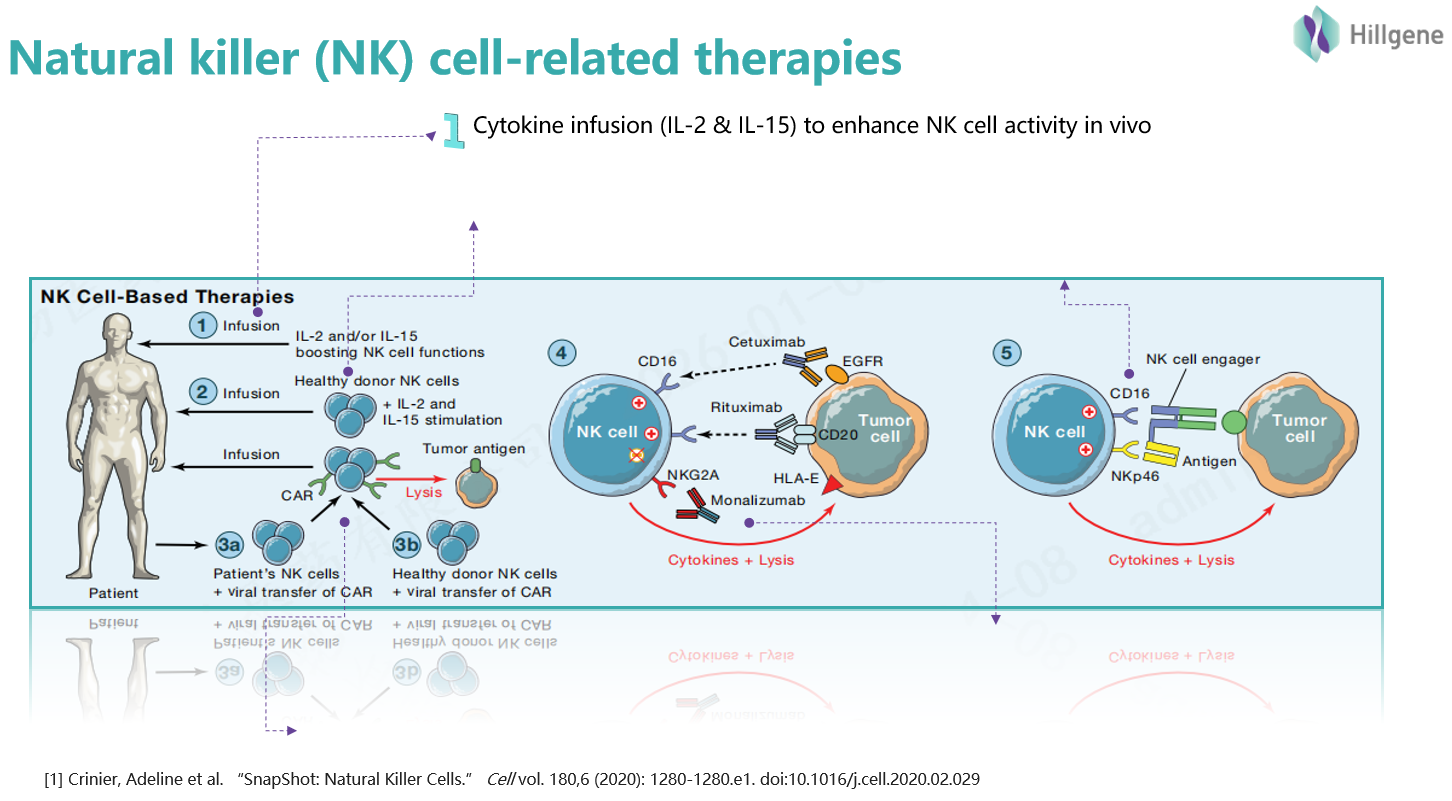
The delayed market entry of NK cell therapies stems from three core challenges: expansion efficiency, in vivo persistence, and cytotoxic activation. The process for large-scale in vitro expansion of NK cells is highly technical. An increasing number of “off-the-shelf” cell therapy pipelines are adopting the feeder cell method (which offers high expansion efficiency but involves a more complex process), while others opt for the feeder-free cytokine method (which features a simpler process but generally yields lower expansion rates). Additionally, the immunosuppressive microenvironment of solid tumors and the short in vivo survival time of NK cells are key factors limiting therapeutic efficacy.
III. The Next Three Years: Who Will Cross the Finish Line First?
Based on clinical progress and regulatory trends, the industry widely predicts that the first approved NK cell therapy is likely to emerge in the United States or China within the next three years. Leveraging the FDA’s accelerated approval pathway and its accumulated early-stage clinical data, the U.S. may be the first to approve CAR-NK products for relapsed/refractory hematological malignancies (such as acute myeloid leukemia and multiple myeloma). While the U.S. relies on the FDA’s policy fast track for its sprint in the field of hematological malignancies, China is building its own “NK cell therapy fast track” by leveraging the vast potential for real-world data translation and the cluster advantages of its upstream and downstream industrial chains. From early-stage R&D to commercial production, Chinese companies are demonstrating faster clinical advancement and stronger cost control capabilities. The largest-scale clinical applications and the most groundbreaking expansion of indications are highly likely to occur in China.
Yingbairui’s IBR854 (targeting 5T4) and Dabo Bio’s E10H (autologous NK) have entered Phase II clinical trials, becoming among the first products in China to race toward market approval. Xianbo Biotech’s SNC103, as the first allogeneic CAR-NK to enter clinical trials in China, is expected to advance rapidly in the field of hematological malignancies if Phase I data prove promising, leveraging the CD19 target and potentially benefiting from policy support due to its “first allogeneic” status. Meanwhile, Zhongsheng Suyuan’s NCR300, as China’s first iPSC-derived iNK to receive clinical approval, offers cost advantages at scale through its technology platform. If subsequent expansion into new indications proceeds smoothly, it could become a blockbuster product after 2028.
Notably, the number of IND approvals in the NK cell therapy sector is projected to grow significantly between 2024 and 2025. In the long term, with technological advancements and process optimization, NK cell therapy is expected to enter a period of explosive commercialization around 2030. Over the next 5 to 10 years, as production processes continue to improve and the industry chain matures, the accessibility of NK cell therapies will gradually increase. By then, NK cell therapies will no longer be a “high-end, bespoke” treatment accessible only to a select few, but will instead become a widely available, inclusive treatment option, bringing new hope to hundreds of millions of cancer and autoimmune disease patients worldwide.
Based on clinical and regulatory trends, industry experts anticipate that the first approved NK cell therapy drug may emerge in China or the United States within the next three years. The U.S. is expected to leverage the FDA’s Fast Track designation to be the first to launch a CAR-NK product targeting relapsed or refractory hematologic malignancies. Meanwhile, China, drawing on its vast real-world data and comprehensive industrial chain advantages, has demonstrated outstanding performance in clinical advancement, mass production, and cost control, positioning it to achieve the largest-scale clinical application and breakthroughs in indications for NK cell therapy.
Several key domestic products have entered critical clinical stages: Yingbairui’s IBR854 and Dabo Bio’s E10H have advanced to Phase 2 clinical trials; Xianbo Bio’s SNC303 is China’s first allogeneic CAR-NK; and Zhongsheng Suyuan’s NCR300 is the first iPSC-derived iNK, all of which possess blockbuster potential.
IND approvals in the NK cell sector are projected to grow significantly in 2024–2025, with the industry expected to enter a commercialization boom around 2030. Over the next 5–10 years, as manufacturing processes mature and the industrial chain matures, NK cell therapy will transition from high-end customization to widespread accessibility, bringing new hope to hundreds of millions of cancer and autoimmune disease patients worldwide.
[1] Wang M, Zhou T, Liu S, Xiang W, Xie K, Zhang X, Hu W, Fang M, Zhang Z, Chen M, Wang X, and Wu J (2026) Global Panoramic analysis of clinical research in cell therapy: clinical trial landscape, marketed products, and regulatory trends. Front. Pharmacol. 17:1715984. doi: 10.3389/fphar.2026.1715984